What is Pharma 4.0
Technology writer, Alasdair Gilchrist, describes Industry 4.0 as the convergence of people, physical systems, and data within an industrial process to increase quality, productivity and profit by using the power of advanced data analytics. Pharma 4.0 can be defined in the same way. Our Pharma 4.0 consultants can help you understand what is Pharma 4.0 and how it is applicable to your organisation.
Why 4.0? It’s a play on two things:
- the numbering system used in software development, and
- the idea that the Industrial Revolution, characterised by mechanisation and steam power, was Industry 1.0; the arrival of electricity was Industry 2.0; the Internet was Industry 3.0; and we are now in the infancy of Industry 4.0 (and Pharma 4.0).
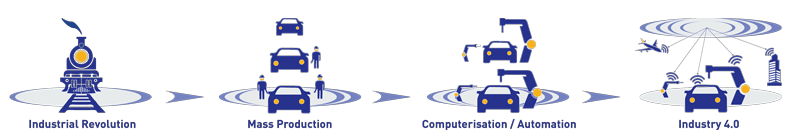
Our understanding of Pharma 4.0
To leverage off modern computing power, a digital twin needs to be created, this digital twin can be an extremely small element of a system or extremely complex. Take the obvious example of an autonomous car, cameras need to continually scan the environment, assessment hazards, road, weather conditions and then safely navigate through the twin digital world. In a pharmaceutical setting, critical GMP records would need to be retained and the automated systems, validated.
Pharma 4.0 Consultants
Our Pharma 4.0 consultants can help with your Industry 4.0 initiatives in Big Data, Additive Manufacturing (we have been consulting 3D printing companies since 2006) and the Industrial Internet of Things (IIot), all referred to as Industry 4.0, and how it’s changing the face of industries like transportation and logistics, manufacturing, aviation, and oil and gas production. But not so much has been written about how Industry 4.0 solutions can be applied to pharmaceutical manufacturing problems to increase quality and productivity, and decrease risk and waste. Pharma 4.0 is the term used to describe Industry 4.0 in a pharmaceutical manufacturing setting.
PharmOut are experts in Quality By Design, balancing the regulatory requirements and innovation, PharmOut’s Pharma 4.0 consultants can assist you with you next project. Whether its a existing plant or designing a state of art facility from scratch, we can help.
To learn more about Pharma 4.0 contact us or read Pharma 4.0 blogs and watch our events as we frequently offer events on Pharma 4.0.

