Benefits of PIC/S Membership for Regulators and Industry
On 1 January 2021, ANVISA, Brazil became the 54th member of PIC/S. Like all other PIC/S members, both ANVISA and Brazil’s pharmaceutical industry will realize a wide range of benefits of PIC/S membership, particularly at this time of the Covid-19 pandemic when on-site GMP inspections of local and foreign sites have been curtailed substantially.
The current membership of PIC/S is shown in the world map below.
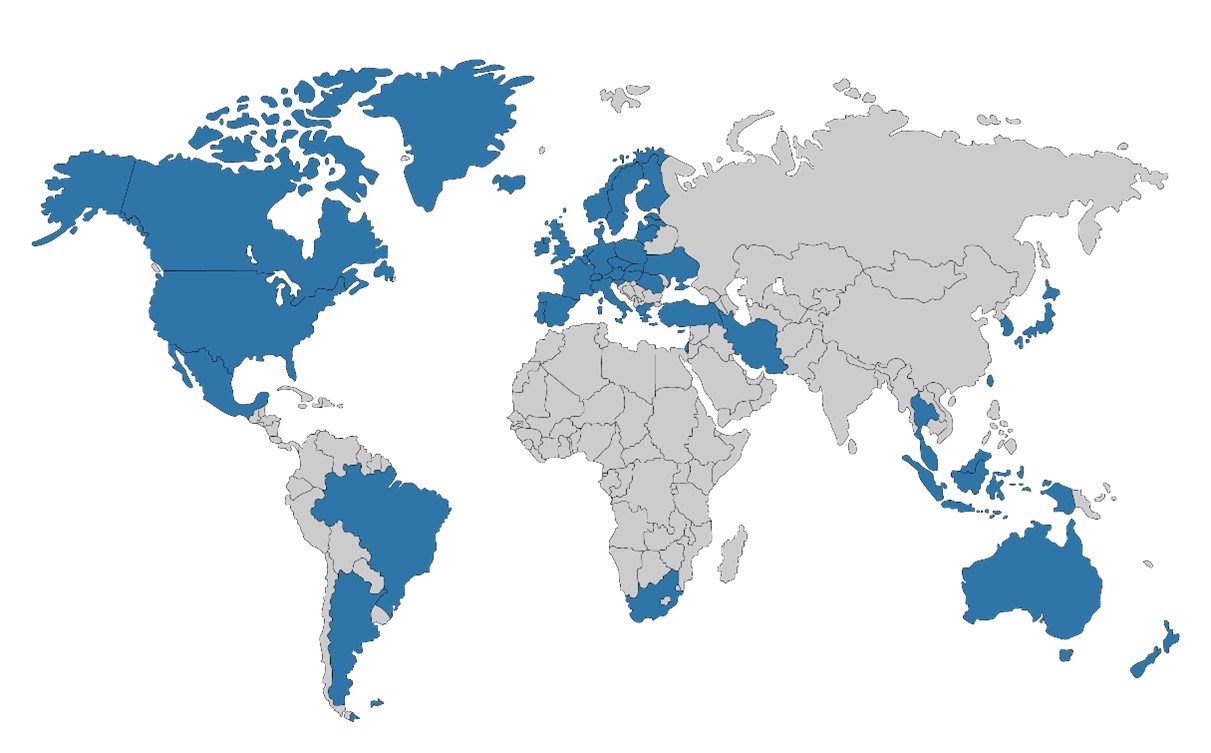
Over the past 20 years, the number of PIC/S participating authorities has increased from 23 to 54. Currently, there are 5 applicant authorities, and 3 pre-applicant authorities are in the process of preparing for PIC/S membership. (See table below).
PIC/S Applicants & Pre-applicants
It usually takes about 3 to 4 years for an applicant authority to become a member of PIC/S. The pre-application process offers a regulatory authority the opportunity to determine, using a gap analysis, whether they are ready to lodge a full application.
Benefits of PIC/S Membership for Regulatory Authorities
In order to become a member of PIC/S, a regulatory authority must make a wide range of improvements to its legislation, inspection procedures, Quality System, inspector training, etc. to achieve equivalence with the systems and procedures of current PIC/S member authorities. The main criteria for PIC/S membership are contained in a checklist setting out the basic systems, procedures and legislation that must be in place in order to obtain membership. This process forces an applicant authority to elevate its systems and procedures to an internationally harmonized level and, once becoming a member, the need to maintain this elevated standard.
Moreover, for a PIC/S member authority, there are many other ongoing benefits, including:
- The sharing amongst member authorities of inspection reports on GMP inspections undertaken, using the PIC/S GMP Inspection Reliance initiative. This is particularly relevant at this time of the Covid-19 pandemic when regulatory authorities are unable to travel to conduct foreign inspections on-site.
- There are significant cost savings that result from this information sharing because there is a much reduced need to travel overseas to inspect foreign sites and to rely instead on the inspection reports of other PIC/S member authorities. It is now common during the pandemic for a PIC/S member authority to carry out a remote desktop inspection of a foreign facility which may include a request, as additional information to make a determination on the GMP compliance status, for any inspection reports from other PIC/S member authorities that may have undertaken on-site inspections of the foreign facility.
- The ability to participate in the development and use of internationally harmonized guidelines on GMP inspection practices. For example, PIC/S is likely to prepare a guideline document on “Remote Inspections” which have become important during the time of the Covid-19 pandemic.
PIC/S held a virtual training Seminar for GMP inspectors in December 2020 on “Distant Assessment of GMP Compliance”. The usual outcome of such Seminars is for a Working Group to be established to prepare a guideline document related to the Seminar topic.
- The opportunity for the training of GMP inspectors, e.g., New Inspector Training, annual PIC/S Seminar on a specific GMP topic, Expert Circle training on a specific GMP topic, Joint Visits Program and on-line training webinar through the PIC/S Inspectorates’ Academy.
- Attaining international recognition that the regulatory authority is using an internationally harmonized GMP requirements and inspection procedures.
Benefits of PIC/S Membership for Industry
The pharmaceutical industry of the country in which the PIC/S member authority is regulating also benefits, mainly from increased opportunities to export pharmaceutical products, not only to PIC/S member countries but also to non-PIC/S member countries (which hold PIC/S inspection standards in high regard).
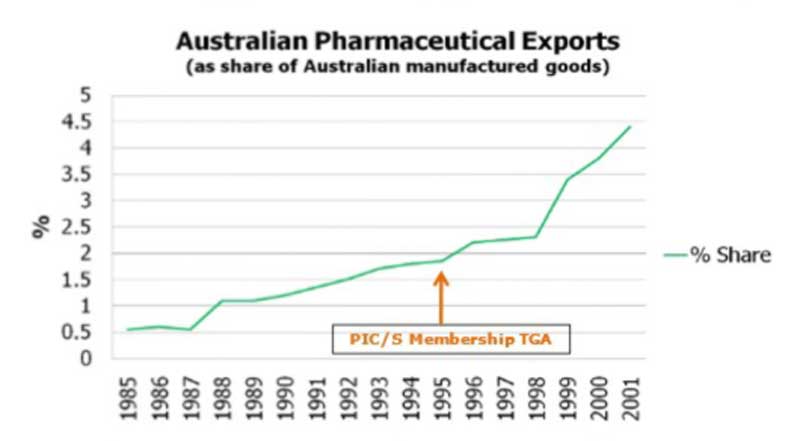
When TGA, Australia became a member of PIC/S in 1995, there was a significant growth in Australian pharmaceutical exports all around the world in the years that followed.
Growth in Australian Pharmaceutical Exports Resulting from PIC/S Membership
Mr George Messinis, Centre for Strategic Economic Studies, Victoria University of Technology, Melbourne
The other significant benefit for industry is the reduced duplication of same-scope GMP inspections and the cost savings that result, particularly during the Covid-19 pandemic when PIC/S member authorities are forced to rely more on the sharing of inspection reports and other information related to GMP inspections. It is possible that this approach will continue after the pandemic ends, albeit in hybrid form. This is consistent with the intent of the PIC/S GMP Reliance initiative.
Before the pandemic, it was common for many pharmaceutical companies to be subjected to numerous same-scope GMP inspections by different regulatory authorities, which was a significant cost burden for industry and resource-intensive for regulators. It has been reported that inspected companies need 10 times the resources of regulators for inspection preparation and conduct, as shown in the following examples (Annual Regulatory GMP/GDP Inspection Survey 2017 Data):
The PIC/S GMP Inspection Reliance initiative and the possible continuation of remote inspections in hybrid form would help reduce the need for duplicate, same-scope GMP inspections after the end of the pandemic.
Read more about PIC/S and the impact of Covid -19 on the Pharmaceutical Manufacturing Industry
Bob Tribe assisting Saudi Arabian FDA application for PIC/S membership


